
News
Science: Phosphate recovery from animal manure: closing nutrient cycles and improving soil health
ETE PhD scientist Chris Schott managed to efficiently recover phosphate from animal manure by adapting ETE’s phosphate recovery technology developed for human toilet waste. The first pilot plant of 5 m3, recovers about 70 percent of phosphorus from animal manure.
Phosphorus (P) is an important and finite natural resource, essential for agriculture as fertilizer. About 70 percent of the natural P reserve that can be used in agriculture is present in phosphate rock in North Western Africa. However, this resource is slowly running out, while P recovery and recycling is at present very limited. In The Netherlands, 71 million kilograms of this nutrient are annually ending up in mainly cow and pig manure. Part of it is reused by farmers to fertilize their crops, but legal limits apply to avoid excess phosphorus to wash out from the soil, harming the environment. As a result, there is a substantial manure surplus, that often is incinerated or transported to other countries, at high economic and environmental costs, since it contains about 90 percent of water. ‘Too much P is wasted at high economic and environmental costs, while without this nutrient, we can’t grow any crops’, Schott says. ‘To close nutrient cycles and guarantee food safety, P-recovery from manure is essential.’
Coincidentally discovered
The method Schott is using to recover P was coincidentally discovered by PhD student Taina Tervahauta some years ago. She studied ways to increase the efficiency of P recovery from human waste using a traditional anaerobic digestion reactor. In this reactor microorganisms convert organic matter into biogas (methane). She treated separately collected toilet water, also called ‘black water’, from a demonstration neighborhood in Sneek, equipped with vacuum toilets. After running the reactor for a year, she was surprised to find small granules with a solid core at the bottom of the reactor. The number of granules slowly increased over time from the bottom up and eventually took over the sludge bed. She found similar granules also at the demonstration site in Sneek and realized this might be something new. The scientist started to sample the mysterious granules to determine their composition and found that they mostly consisted of pure calcium phosphate crystals. That was a great find, since these crystals were not only very pure, but could also easily be harvested for reuse because of their size.

Challenges
Now a new method for P recovery was established, scientists further developed and improved the microbial formation of calcium phosphate crystals from human waste at Water Research Institute Wetsus in Leeuwarden. By adding extra calcium, scientists were able to recover as much as 90 % phosphorus as calcium phosphate granules using the anaerobic digestion reactor. Schott further adapted the method for P recovery for both pig and cow manure. However, there were several challenges that needed to be met.
In pig manure, the scientists had two main problems. Due to the high amounts of the toxic ammonia present, anaerobic digestion was inhibited, although calcium phosphate granules were still formed. A bigger problem was the high fraction of solids present in pig manure, making it too thick to process. ‘These solids mainly consisted of organic material’, Schott explains. ‘By separating these solids from the nutrient-rich liquid fraction using a screw press (fig. 1), we came a step further, but still reactor pipes were clogging due to many small particles also as a result of adding extra calcium, resulting in more precipitates.’ By simply using a bigger reactor with bigger pipes, that problem could be solved, and after adding the extra calcium needed, this resulted in a P recovery of almost 75 percent.
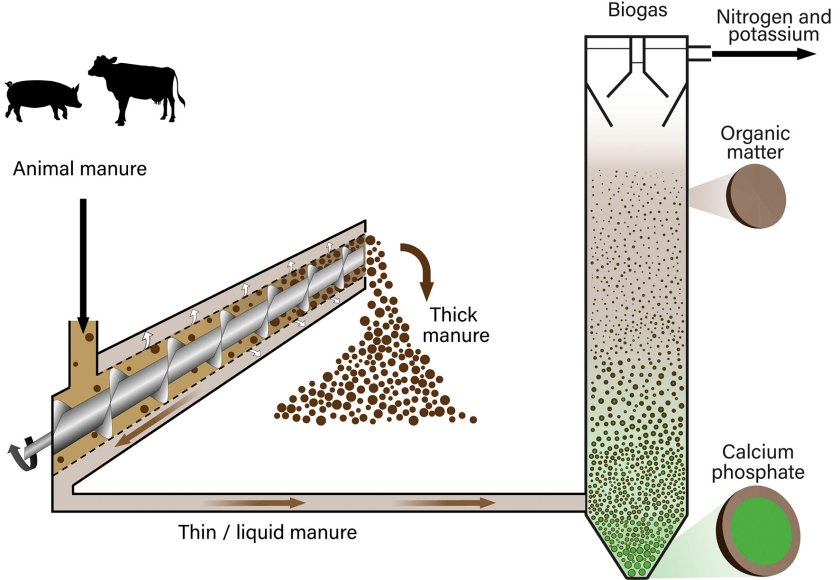
Cow manure posed some different problems: after separating the liquid and the solid fractions, there was still a high amount of small fibers present, absorbing a lot of the calcium added, resulting in a lower P recovery and smaller crystals. Still, P removals of 70% were possible, but the recovery, reuse and transportation were more difficult. ‘We solved this problem by pretreating the cow manure with a lower pH, that gets rid of some of the fibers present’, Schott explain. ‘Also, this resulted in overall bigger calcium phosphate granules.’
Improve soil health
The adapted UASB method has resulted in a pilot plant of about 5 m3, that retains about 70 percent of phosphorus present in animal manure, but the scientists believe 90 % is feasible. The whole separation process results in an efficient reuse of organic materials and nutrients: farmers can use the solid organic fraction separated in the first step to improve soil health. The organic matter remaining in the more liquid fraction is converted into biogas in UASB reactor.
However, in addition to P, all the nitrogen and potassium are also within this liquid, which allows a separate collection and application of phosphorus from nitrogen and potassium. This can be very useful to dose different amounts of these nutrients but also to fertilize with different timings, since optimal doses and timings differ for crops but also grasses. So, no incineration or transport of raw manure is needed anymore. This approach increases the value of animal manure and stimulates circular agriculture.

Selected publication:
Cunha J.C., Schott C., van der Weijden R.D., Leala R.H., Zeeman G., and Buisman C.J.N. 2020. Calcium Phosphate granules recovered from black water treatment: A sustainable substitute for mined phosphorous in soil fertilization. Resour., Conserv. and Recy. 158, 104791